The LNB straddles the interface of nanotechnology and protein engineering. Equipped with mechanisms of unparalleled molecular recognition and autonomous fault-tolerance, biological constructs offer cost-effective approaches for the manufacturing of intricate molecular motifs. The commercial development of protein-based constructs for light-harvesting and biosensing applications, however, has been hindered by protein instabilities due to photodegradation and photobleaching. Inorganic nanomaterials, on the other hand, demonstrate enhanced photophysical and electronic properties that enable optical devices with unprecedented, quantum optoelectronic properties.
In a sense, the advantages and disadvantages posed by the fields of protein engineering and nanotechnology are highly complementary. The LNB focuses on exploiting the synergy between these two fields in engineering a new generation of nano-bionic devices.
Biosensors
Single-walled carbon nanotubes (SWCNTs) are rolled up sheets of graphene that form a tube of carbon atoms. Upon laser excitation, semi-conducting SWCNTs fluoresce with emissions in the near-infrared range of the optical spectrum. These near-infrared wavelengths are emitted within an optical “window” that lies above blood and below water absorption, making SWCNTs ideal for in vivo applications that benefit from minimal background and tissue interference. SWCNT fluorescence is strongly modulated by the nanotube’s immediate environment.
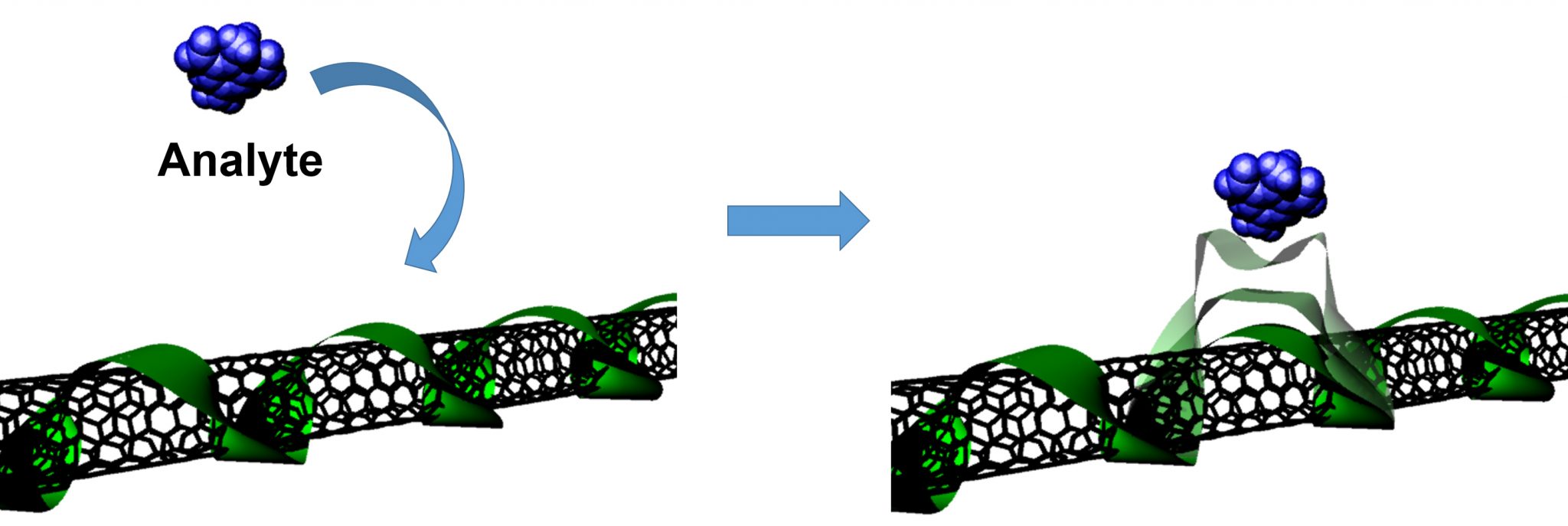
SWCNTs (black) are wrapped with surfactant, polymeric, or biological coatings (green). Binding to an analyte (blue) changes the immediate environment of the SWCNT, resulting in a change in fluorescence emission (Nature Nanotech. 2013).
Every carbon atom of the SWCNT is exposed to its surrounding environment, imparting the SWCNT with fluorescence sensitivities that extend down to the single-molecule level. In other words, a SWCNT can sense individual molecules reversibly binding to its surface. These binding events affect the nanotube fluorescence, causing the nanotube to blink as it interacts with individual molecules!
 |
 |
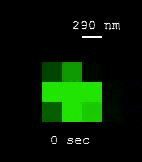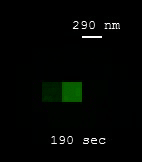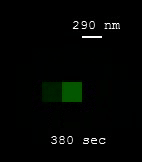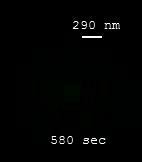
| [MOVIE] Individual molecules of nitric oxide (NO) reversibly binding to the nanotube surface results in the blinking of SWCNT fluorescence (J. Am. Chem. Soc. 2011) |
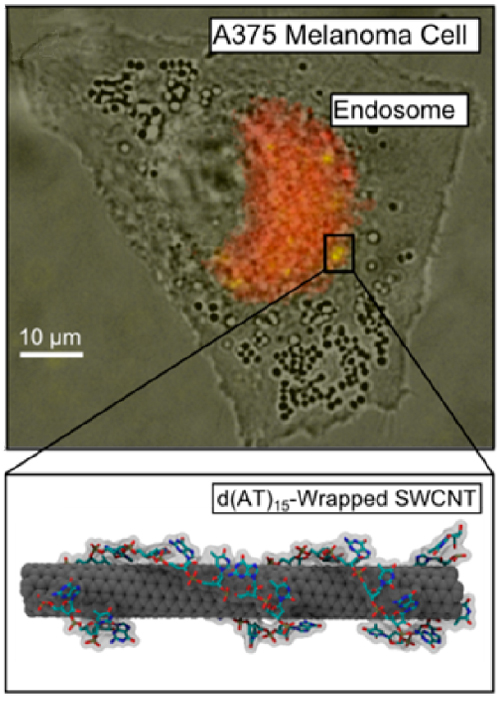
d(AT)15-wrapped SWCNT sensors inside an A375 melanoma cell image the spatiotemporal distribution of NO within the cell in real-time (Nano Lett. 2014) |
.
The reversible binding events on the SWCNT surface remains largely non-specific. By coupling the SWCNT surface with biological and engineered constructs with enhanced molecular specificities, the LNB offers an interdisciplinary approach towards engineering these nanosensors for in vivo and protein engineering applications.
Solar Energy Conversion
Photosynthesis is the process by which plants convert solar energy to chemical energy. Water and carbon dioxide (CO2) are taken up by the plant. The absorbed light is used to split water (an energy intensive process!) to produce oxygen, and the CO2 is incorporated into sugars. These photosynthetic systems are predicted to have maximum energy conversion efficiencies of 26.8%, comparable to the 31.8% efficiencies predicted for commercially available, single-junction cells. Advantages in fault tolerance, self-repair mechanisms, material abundance, material cost, and CO2 sequestration make biological materials attractive for solar energy applications.
Despite these advantages, biological photovoltaics (PVs) remain an underdeveloped technology hindered by poor energy extraction and protein stability mechanisms that may limit their overall efficiencies. The LNB leverages techniques in synthetic biology and protein engineering to engineer light-harvesting, biological constructs with enhanced synthetic activities; proteins and living cells are being “re-programmed” to behave like, and even interact with, synthetic devices.
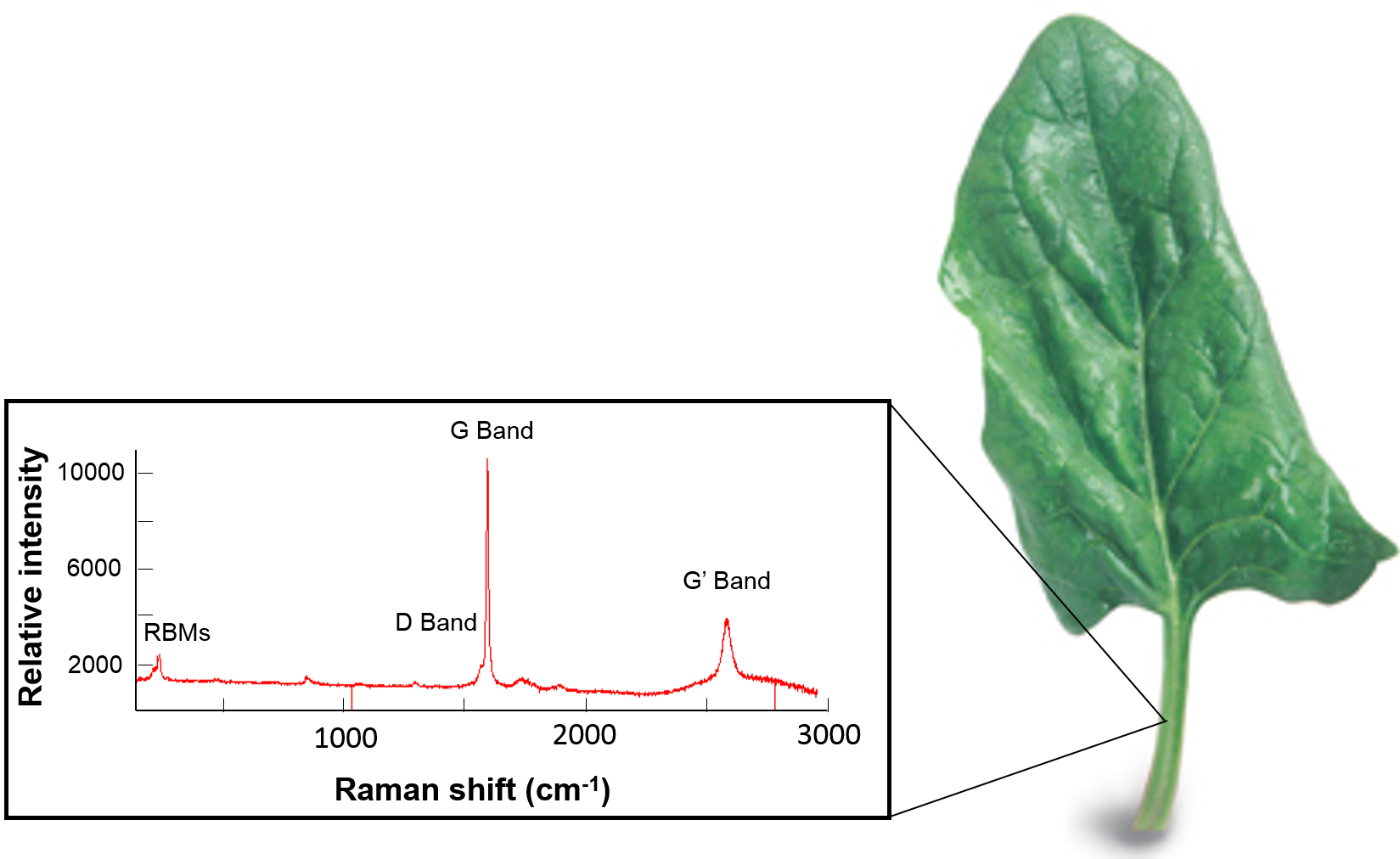
Polymer-wrapped SWCNTs are taken up by a spinach leaf (right) for in vivo sensing and energy applications. A raman spectrum of the leaf (left) confirms the expected optical SWCNT signature (Data unpublished, 2008).
.
 |
By expressing 3 foreign proteins in E. coli, the recombinant cells are able to transport electrons through their otherwise insulating outer membranes. Such recombinant cells have been interfaced with electrodes to create hybrid biosensing and energy devices. The bioreactor shown here (left) contains recombinant E. coli that consume sugar to produce electricity. |
.
The ability engineer both proteins and nanoparticles to augment biological functions allows the LNB to create hybrid, nanobionic devices with advantages that lie beyond what could be achieved in the natural and synthetic worlds alone.
.
 |
 |
.
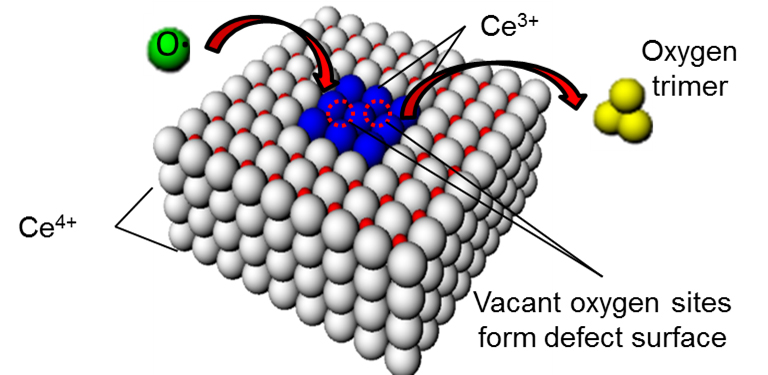
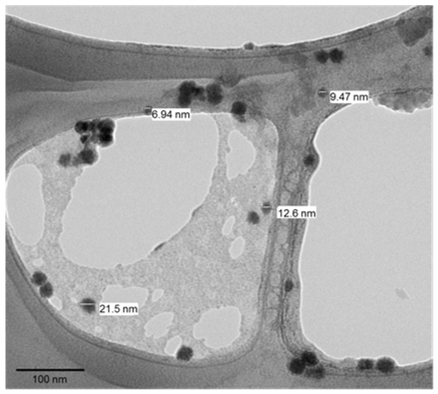
Cerium oxide (CeO2) nanoparticles, or nanoceria (NC) (left), regeneratively scavenge for damaging reactive oxygen species (ROS) (green) that are generated during photosynthesis. Transmission electron microscope (TEM) images (right) confirm NC size. NC addition to intact chloroplasts has been shown to enable hyperstable chloroplasts for solar energy harvesting (Adv. Energy Mater. 2013).
Wastewater treatment
Presentation of the microbial wastewater treatment at LNB
Food industry
Presentation of the Future Food Initiative partnership with LNB
Bioengineering for Electron Transfer
Presentation of Mohammed about the project